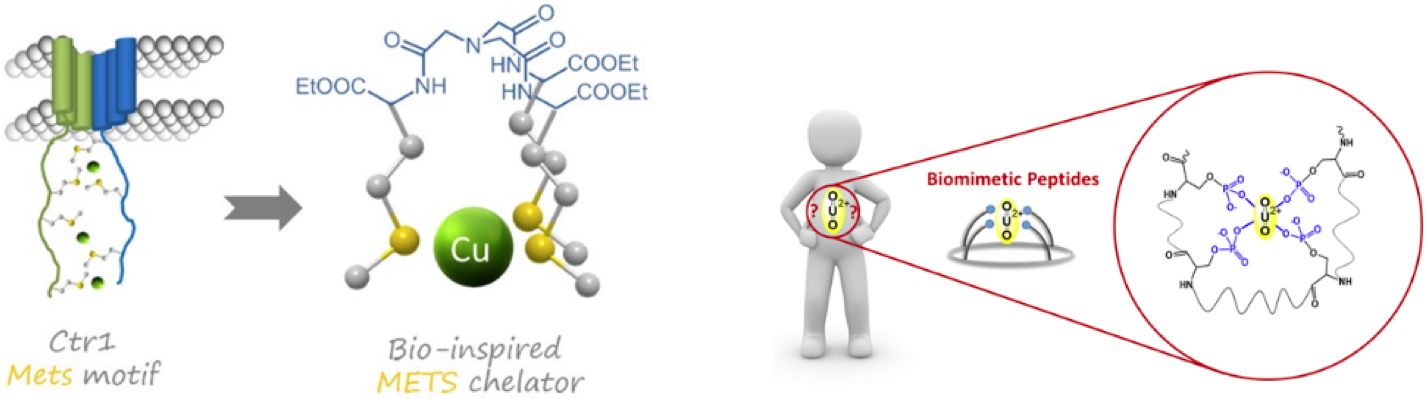
Pascale Delangle & Sarah Hostachy In living organisms, metals can interact with a number of biomolecules, including metabolites, lipids, and proteins. They generally interact with proteins through their amino acids side chains or the peptidic backbone. Essential metals can act as structural cofactors, by facilitating the spatial arrangement of a set of amino acids, or trigger conformational change in the protein. They can also be metabolic cofactors in the catalytic site of metalloenzymes. On the other hand, xenobiotic metals can compete with essential metals, interfere with protein function through change of structure or function, and cause oxidative damage. In either case, these interactions highly depend on the metal species and the nature of amino acids involved in the interaction. The understanding of these interactions is thus critical for the further development of bio-inspired medication or
detoxifying agents.
Our approach relies on the careful design of small peptides-metal complexes to mimic the protein-metal environment. Indeed, proteins comprise several hundreds of amino acids, but only a handful of them interact with the metal species. In addition, proteins are often more sensitive to environmental factors (pH, temperature, solvent, ionic strength, etc.). Small peptide or peptide-like molecules that reproduce the metal-protein environment are thus particularly well adapted to dissect the mechanisms of metal-protein interaction. They are generally more robust than proteins to experimental conditions, and their properties can be readily tuned by varying the peptide sequence or introducing unnatural amino acids during the synthesis.

Related publications
|
Mesterházy E, Lebrun C, Jancsó A and Delangle P A constrained tetrapeptide as a model of Cu(I) binding sites involving Cu4S6 clusters in proteins.
Inorganic Chemistry, 2018,
57(10): 5723–5731 |
Mesterházy E, Lebrun C, Crouzy S, Jancsó A and Delangle P Short oligopeptides with three cysteine residues as models of sulphur-rich Cu( I )- and Hg( II )-binding sites in proteins.
Metallomics, 2018,
10(9): 1232–1244
Laporte F. A, Lebrun C, Vidaud C and Delangle P Phosphate-rich biomimetic peptides shed light on high-affinity hyperphosphorylated uranyl binding sites in phosphoproteins.
Chemistry – A European Journal, 2019,
25(36): 8570–8578