Scalable synthesis of nanostructured Si based materials remains a challenge. In CVD growth, SiNWs are obtained by reacting a gas flow of silane SiH
4 on a substrate covered with gold nanoparticles Au
NP at >500°C. SiH
4 dissociates on molten gold droplets, and Si atoms dissolve in the metal. At saturation, Si starts to crystallize as a nanowire. Trouble is that SiH
4 is an pyrophoric, explosive gas.
SiH
4 + Au
NP → 2H
2 +
SiNW
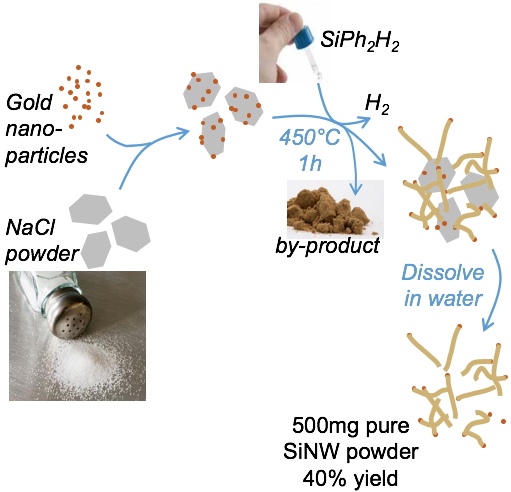
In our process, we switch to a chemist view of SiNW growth:
1/ a yield oriented growth: we use a small reactor volume with high silane and gold loading
2/ we use an organosilane oil as Si source because it is an air stable reagent
3/ reaction is made in 3D for mass production: for this, gold is deposited on NaCl micro-powder. NaCl offers large surface area and keeps catalysts apart. The salt is removed at the end of the reaction by washing with water.
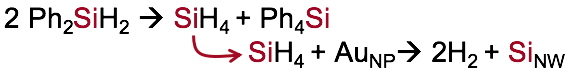

 ReferencesO Burchak, P Chenevier, P Reiss
ReferencesO Burchak, P Chenevier, P Reiss Patent FR1455431, 2014
Scalable chemical synthesis of doped silicon nanowires for energy applicationsNanoscale (2019) 11, 22504-22514
O Burchak, C Keller, G Lapertot, M Salaün, J Danet, Y Chen, N Bendiab, B Pépin-Donat, C Lombard, J Faure-Vincent, A Vignon, D Aradilla, P Reiss and P Chenevier