Published on 13 October 2025
Body text 1
Jean-Luc Ravanat, Marie Carrière & Thierry Douki
Evaluating the impact of toxic contaminations or deleterious environments on human health requires a quantitative assessment of the nature and the severity of the exposure. This is the purpose of biomonitoring, an approach that involves quantification of cellular responses known to be characteristic of specific exposures. These biomarkers can be of various nature, including modulation of gene expression, production of specific proteins or changes in cellular metabolism. Biomonitoring can also rely on the quantification of specific analytes in biological matrices. This strategy is widely developed at CIBEST. We use biochemical assays and chemical analysis tools (
HPLC-MS/MS) in order to quantify damage to biomolecules, in particular to DNA.
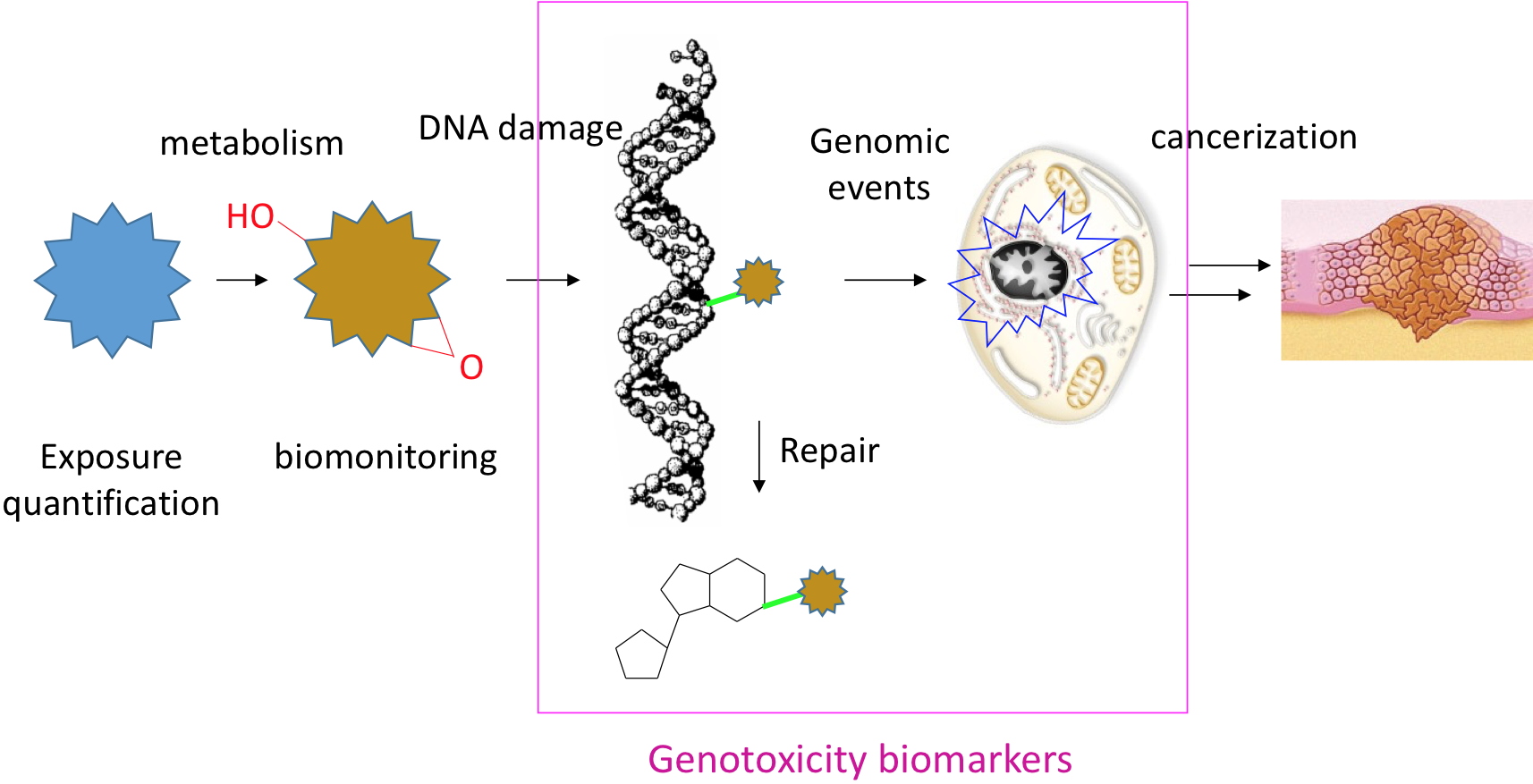
Main steps leading from the initial exposure to a DNA damaging agent to the development of tumors.
Genotoxicity biomarkers studied at CIBEST damage quantified in nuclear DNA, modified lesions released upon repair and genomic events resulting from the presence of damage.
This strategy has been applied to several topics. For example, the quantification of 8-oxo-7,8-dihdro-2’-deoxyguanosine in urine is an easily accessible and sensitive biomarkers of
oxidative stress. In a more recent work, methods were developed at CIBEST for the quantification of the adduct between
sulfur mustard and guanine in cell culture medium and in the plasma of cutaneously exposed mice. Modification products of glutathione, a tripeptide involved in the cellular protection against oxidative stress and electrophilic chemical species, were also quantified in the same samples. A more complex approach uses UV-induced
DNA photoproducts for the evaluation of the protection afforded by sunscreens. This work requires collection of biopsies from exposed volunteers followed by a tedious extraction of DNA from the collected skin. The extent of protection is then determined by comparison of the HPLC-MS/MS data obtained in skins exposed to simulated sunlight with or without sunscreen.
Top page