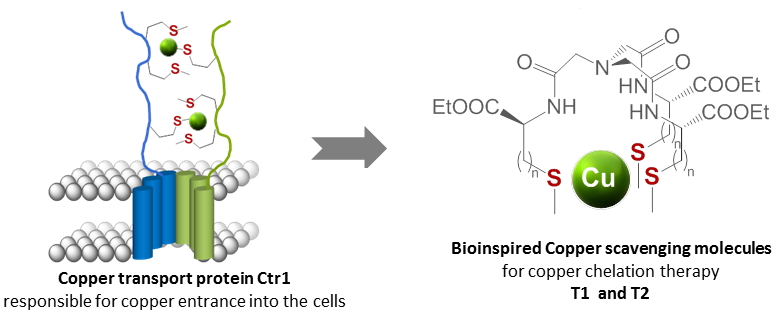
Copper is vital but also toxic for life. It plays an essential role in a number of vital processes, but high concentrations of copper can be deleterious leading to oxidative damage. Proteins involved in Cu regulation are a fascinating source of inspiration in the design of Cu scavenging molecules for copper chelation therapy. Among them, the Cu(I) transport protein Ctr1 is essential for copper entrance into the cells. It binds Cu(I) thanks to three sulfur atoms from three methionine residues.
To mimic this protein, molecular architectures were synthesized based on a nitrilotriacetic acid scaffold functionalized with three amino acids bearing thioether side chains, either methyl cysteine in T1 or methionine in T2. These two ligands T1 and T2 were obtained with good overall yields and demonstrate efficient chelating ability for Cu(I). In each case, only one mononuclear complex, [CuT1,2]
+ was evidenced by electrospray ionization−mass spectrometry (ESI-MS) and the circular dichroism signature showed reorganization of the pseudo-peptide scaffold upon Cu(I) complexation. Considering that thioether functions are neutral sulfur donors, the stability constants are quite large and significantly higher (logK ~ 10) than those evaluated for Ctr1 peptide models (logK ~ 5). This behavior may be assigned to an optimal organization of the sulfur binding sites around the copper ion thanks to the chemical scaffold.
Such Cu(I) chelators are interesting for applications in chelation therapy for treating diseases related to Cu overload, like the rare Wilson’s disease. In addition to their optimal scavenging Cu(I) properties, these ligands are not sensitive to an extracellular oxidizing environment. Furthermore, Cu is also known to be involved in several neurodegenerative diseases such as Alzheimer’s disease and Cu pathways are used to bring anticancer drugs like cisplatin to their target. Therefore the development of such Cu chelators is of great interest for numerous medical applications.
This research project was funded by the ANR émergence, the “Fondation pour la Recherche Médicale” and the labex ARCANE “Chimie Biomotivée”.