This technique applies to many families of nanocrystals and is particularly attractive for non-toxic ones, used in biological applications. With our colleagues from SCIB and Grenoble Institute of Neuroscience, we have developed imaging agents that penetrate the cells, are fluorescent, and increase the contrast in MRI.
Fluorescent semiconductor nanocrystals (or QDs for "quantum dots") offer high performance today: their quantum yield is almost 100%, the emission line is extremely narrow (
i.e. their color is very well defined), and they show excellent photostability. The first bright QDs were based on cadmium, a toxic metal, but nowadays more "biocompatible" systems such as InP/ZnS core/shell nanocrystals are synthesized, which are the subject of this work. All QD features sound very attractive for biotechnologies, except for the (big) detail that the synthesis occurs in organic media. To transfer QDs into aqueous media, the environment of the living, we must replace the hydrophobic surface ligands of the synthesis by hydrophilic ligands. Simple No! Known protocols lead to two major flaws: i) the solutions are not stable, ii) 90% of the fluorescence is lost.
Colloidal stability For a good use of QDs it is essential to keep them in colloidal suspension and avoid aggregation. This is the role of the ligands covering their surface. The thiol function (-SH) is conventionally used, particularly for anchoring on metals such as gold. But in the case of QDs with a ZnS shell, bonding with the thiolate function (-S-) is much stronger. Therefore, the ligand has to be deprotonated, which is done by precisely controlling the pH. For the cysteine ligand, the optimum value is pH 9.2. With this protocol, we increased the colloidal stability of the QDs from one day to several months (Figure 1).
The second problem that we solved is the loss of luminescence during the exchange with cysteine. This problem, already encountered in previous works that operated the ligand exchange in acid medium, is accentuated in basic medium. We found out the main mechanism and how to avoid it: Cysteine has a strong tendency to form dimers containing an S-S bond known to kill fluorescence. If we add a phosphine (TCEP), which selectively reduces this bond, the fluorescence is recovered (Figure 2). Even better is to use TCEP directly in the phase transfer step, which virtually eliminates the problem. The protocol developed for InP/ZnS is equally valid for CdSe QDs, with 0, 1 or 2 shells, for nano-rods, etc.
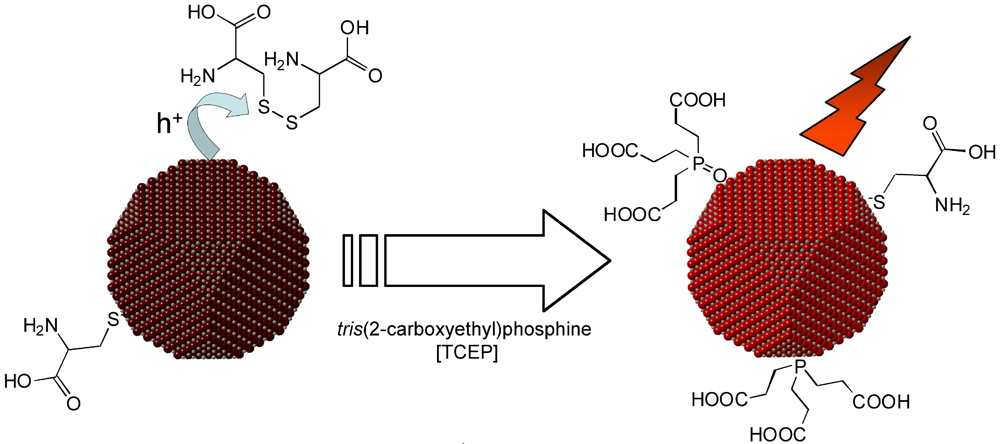
Figure 2: An undesirable side effect of the phase transfer in basic pH is the formation of a dithiol bond between two cysteine ligands. The S-S bridge is a trap for photogenerated positive charges in the nanocrystal, which are no longer available for fluorescence. An efficient remedy is the addition of the reducing agent TCEP. In addition to its reducing action, it may also serve as a ligand for passivating the nanocrystal. With this treatment, more than 90% of the initial fluorescence in chloroform is retained in water.
Probes for imaging Cysteine is a small molecule, so that the size of the nanocrystal with its ligands is less than 10 nm, an advantage allowing the natural excretion of the QDs. We made multimodal probes by grafting other functions, such as rare earth chelates. Among them, gadolinium is an excellent contrast agent for MRI. Our optical / magnetic probes significantly improve the performance of commercial products: higher contrast due to their high relaxivity and retention times extended to several hours (Figure 3a, b). Other rare earths of interest: europium and terbium, which luminesce in other colors and with characteristic decay times a million times larger than semiconductors. We also managed to co-graft a peptide, maurocalcine (MCa), essential for cell penetration (Figure 3c). All this in one step!
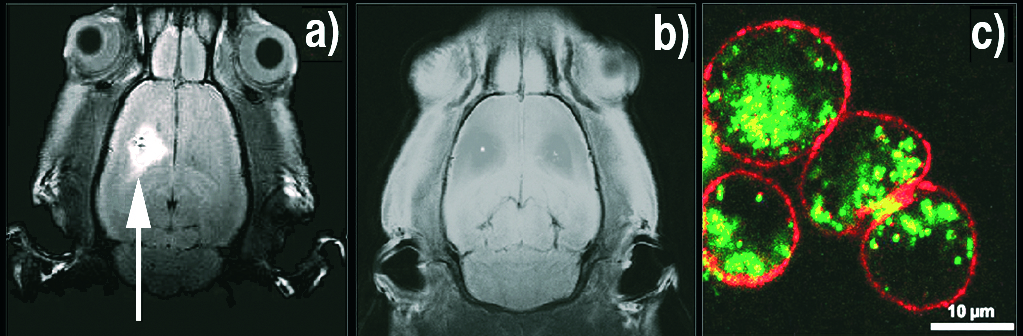
Figure 3: a) False-color view of the presence of contrast agents within Chinese hamster ovary cells. Penetration of the cell does not happen without the presence of MCa peptide, grafted on the fluorescent nanocrystal (green). The cell walls (red) are colored with rhodamine.
b) MRI image of a rat brain 4 hours after injection of the contrast agent Gd-MCa QDs.
c) With the commercial contrast agent Dotarem ®, there is no signal after 4 hours.